
In previous articles, you learned about what a clinical study encompasses and then discovered how to open a file. You have also since read about how compensation is evaluated for each study. Now your file is completed and you are ready to take action. It is now time to choose a suitable study.
It is important to understand how to read a study’s datasheet to know if you are eligible to participate in a particular clinical study. When you visit the webpage of available studies, you will find multiple datasheets that correspond to individual studies:

In this example of a datasheet, important information is presented:
● K65(A)
This is the code attributed to the study. You will need to provide this code to one of our agents should you decide to sign up by telephone.
● Genders accepted for the study
Certain studies are restricted to only men, or only women.
● Compensation
In this example, the compensation offered is $1030.
● The dates for overnight stays at the clinic
This example study has 2 stays of 2 nights each.
● The number of return visits per period
These are obligatory follow up appointments at the clinic, following
the end of a stay. The reasons for these visits are to run additional
blood tests or for medical supervision. In this example, there is a
return visit at the end of each overnight stay, or one per period.
● The no smoking sign
The presence of this symbol implies you must be a non-smoker (or an ex-smoker) to
participate in this study.
● The age of the participants
In this example, the participants must be between the ages of 18 and 65 years old.
When you click the button “See Details and Apply,” you will view additional details regarding the study.
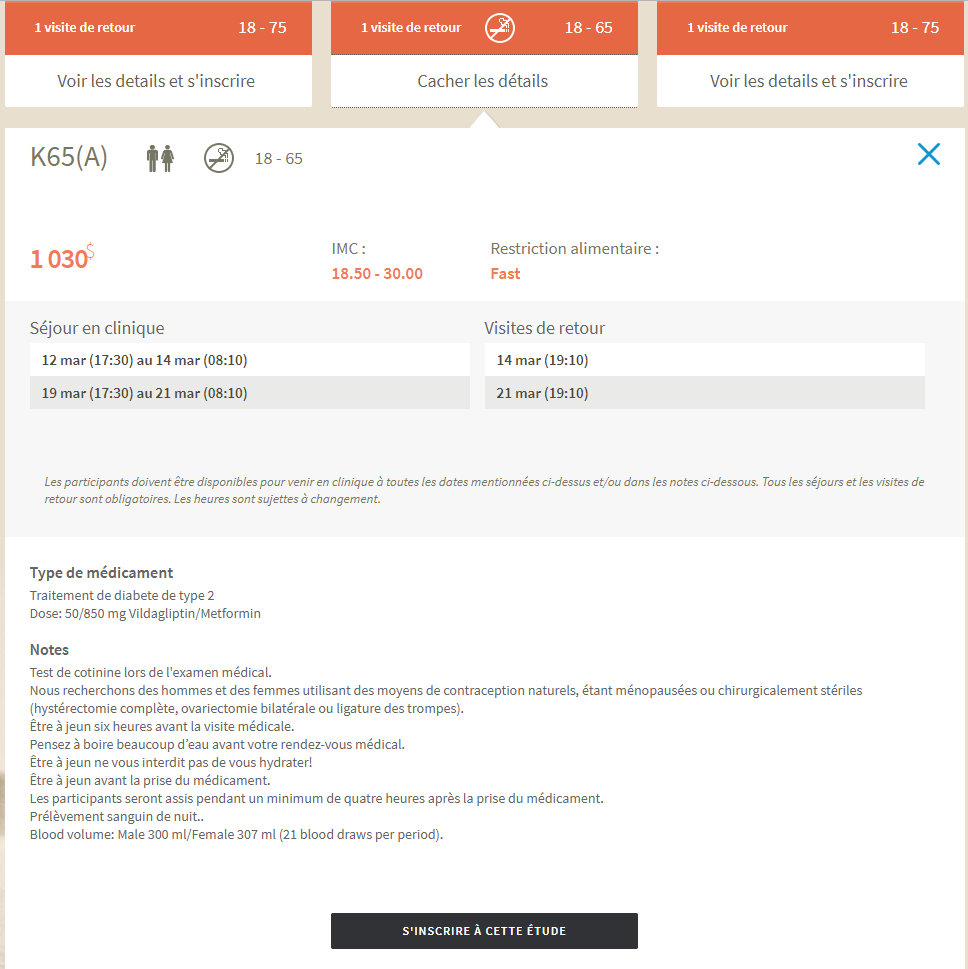
Additional information is then displayed:
● The arrival and departure times for overnight clinical stays
In this case, they are 5:30pm and 8:10am respectively
● The exact dates of the return visits
Additional details regarding the specific admission criteria is also presented:
● BMI (Body Mass Index)
For this study, your BMI must be between 18.5 and 30. If you know
your height and weight, a simple online search will explain how to
calculate your BMI.
● Medication Type
A description of the pathology is given, as well as the dosage and
name of the medication. In this case, a dose of 50/850 mg of
Vildagliptin/Metformin will be administered, with a goal of treating
Type 2 Diabetes.
● Notes
Additional important information to review.
At any time you may consult our guide on how to read a study to better understand the datasheet. The guide is accessible at the top of the available studies page.
When you have decided on a particular study, you may apply for it. You have a choice to click the “Apply for this study” button or to call one of our agents at our call center. Our agents will be happy to answer any of your questions and sign you up for the study.
This now concludes our series “Before the Study”! We invite you to review our “Series Summary”. In the next series entitled “During the Study,” you will have the opportunity to discover how participants are chosen; what kind of environment you can expect during a clinical study; what you can do and bring during a stay and learn more about the medical side of a clinical stay.

